Interaction Risks and Common Escalation Mistakes
The pharmacological basis of high-risk compound combinations, compound-specific side effects of Trenbolone, dose-creep dynamics, evidence-based cardiovascular risk from long-term AAS use, and when to take a meaningful break.
- Explain the pharmacological basis for why stacking two 19-nor compounds is high-risk
- Understand how oral compound hepatotoxicity compounds with multiple orals simultaneously
- Describe the unique mechanism-level side effects of Trenbolone
- Recognise dose-creep patterns and their characteristic behavioural and pharmacological features
- Summarise the cardiovascular evidence base and its practical implications for cycle management
- Apply evidence-based reasoning to recovery cycle decision-making
Stacking Two 19-Nors: The Compounding Problem
Running Nandrolone and Trenbolone simultaneously is one of the most commonly cited examples of compound combination that produces dramatically disproportionate side effects relative to the anabolic benefit. Understanding why requires looking at what each compound contributes at the receptor level, and what the sum of those contributions produces.
Both Nandrolone and Trenbolone share the 19-nor backbone and activate the progesterone receptor with significant affinity. Nandrolone activates the progesterone receptor at approximately 20% of progesterone’s affinity; Trenbolone activates the progesterone receptor at approximately 60–100% of progesterone’s affinity by some binding assays. The combination produces progesterone receptor signaling of an intensity that neither compound generates alone, with compounding effects on prolactin elevation, gynecomastia threshold lowering, and libido disruption via dopaminergic suppression.
The prolactin management challenge becomes substantially harder: both compounds independently contribute to prolactin elevation through dopamine-antagonist-adjacent mechanisms, and Cabergoline must work harder to maintain adequate dopaminergic tone. Some users find that the combination drives prolactin to levels where cabergoline at standard doses becomes insufficient. The gyno risk compounds similarly: the synergistic interaction between progesterone receptor activation and estrogen receptor activation in breast tissue means even carefully managed estradiol may not be sufficient to prevent gynecomastia when both 19-nors are present simultaneously.
Beyond the shared 19-nor risks, the combination adds Trenbolone’s unique side effect burden (addressed in detail below) to Nandrolone’s already-significant cardiovascular and hormonal load. The combined androgenic load from Trenbolone’s 500:500 ratio and Nandrolone’s 125:37 ratio plus whatever testosterone base is running is enormous. The pharmacological case for this combination is weak, both Trenbolone and Nandrolone produce high anabolic activity via the AR; they are largely redundant from an anabolic mechanism standpoint while being actively synergistic from a side-effect standpoint.
Oral Compound Stacking: The Hepatotoxicity Compounding Effect
17-alpha-alkylated oral compounds each generate hepatic enzyme load through the same fundamental mechanism: 17-aa modification forces hepatocytes to process the compound repeatedly without efficient first-pass clearance, generating bile acid accumulation, mitochondrial stress, and oxidative damage proportional to compound concentration and duration of hepatic exposure.
Running two oral 17-aa compounds simultaneously, the most dangerous form being Dianabol + Anadrol for a “mass stack” kickstart, does not produce twice the liver stress; it may produce significantly more than twice, because the hepatic clearance pathways are shared and competing. When two compounds saturate the same metabolic enzymes simultaneously, clearance of both is slowed, effective hepatic concentrations of both rise above what a single-compound analysis would predict, and the hepatocyte stress load is disproportionately amplified.
The combination Anadrol + Dianabol is particularly notorious because both are wet, high-dose oral mass builders with among the highest individual hepatotoxic potential in their class. Experienced users who have run each individually and considered their solo ALT response acceptable are routinely surprised by dramatic liver enzymes elevation when combining them, sometimes reaching levels that require same-day cessation and medical evaluation. The correct approach is sequencing rather than stacking: run an oral kickstart compound at the beginning of a cycle, then discontinue it once long-ester injectables reach saturation, and potentially reintroduce a different oral compound in the final weeks as a finisher, but never simultaneously.
Superdrol (methasterone) deserves special mention because its individual hepatotoxic potential is substantially higher than most other oral compounds even at modest doses. Superdrol produces dramatic ALT elevation at doses as low as 10–20 mg/day in some users, and its hepatic stress profile is considered the worst of widely-used prohormone-era oral compounds. These compounds should be treated as single-compound protocols with strict duration limits (typically 4 weeks maximum), mandatory TUDCA at 500 mg/day, and weekly liver enzymes monitoring if possible.
Trenbolone-Specific Side Effects: A Mechanism-Level Analysis
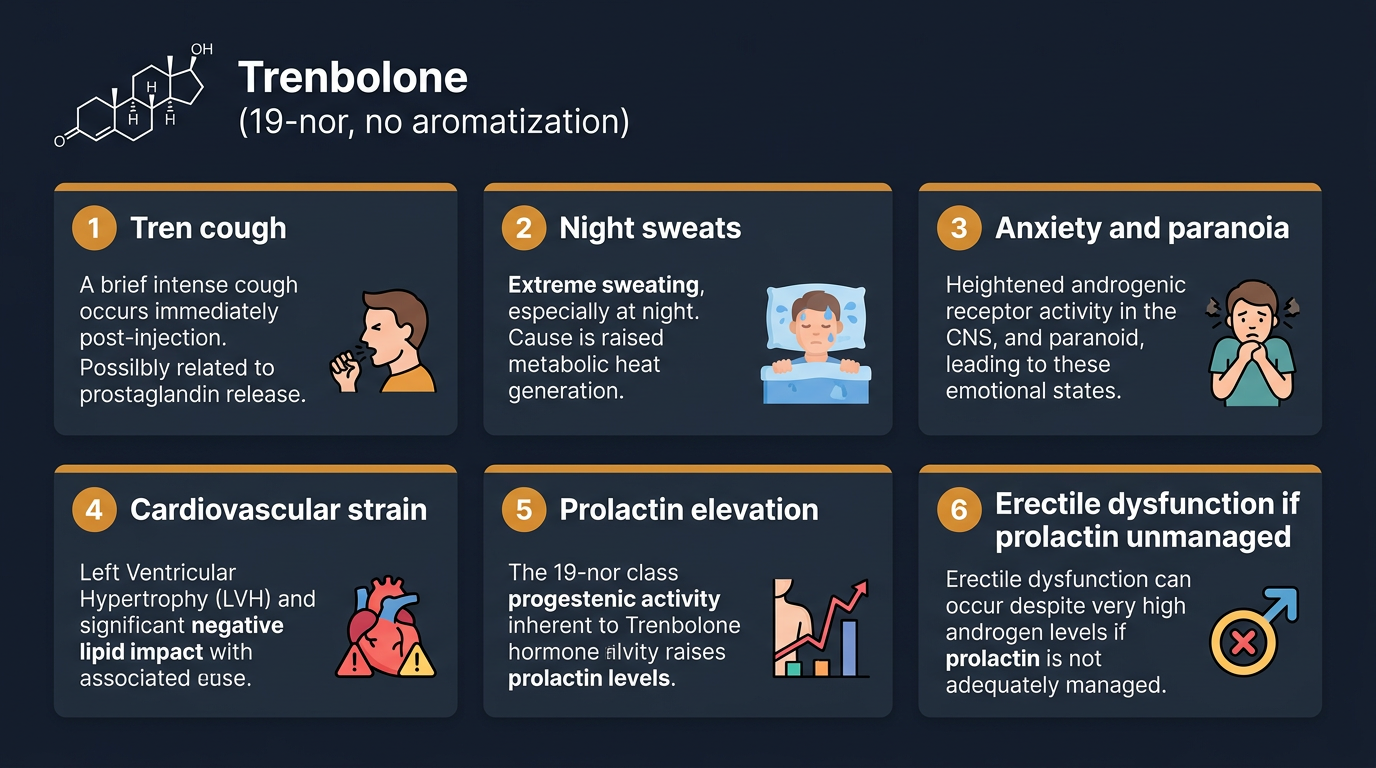
Trenbolone occupies a unique position in the side effect landscape because its effects involve multiple receptor systems simultaneously, not just the AR. This multi-receptor activity produces a side effect profile that is qualitatively different from other anabolic compounds, not merely quantitatively more severe.
Night sweats and thermogenesis: Trenbolone produces significant nocturnal diaphoresis (night sweats) in a majority of users. The mechanism is multifactorial: trenbolone’s glucocorticoid receptor partial agonist activity at certain concentrations alters the hypothalamic thermostat, trenbolone’s effect on catecholamine signaling increases basal metabolic rate and heat production, and the lack of estradiol (from zero aromatization) shifts the heat-dissipation balance. Night sweats are not merely inconvenient, they disrupt sleep architecture, and sleep disruption compounds the mood and cognitive effects of trenbolone.
Tren cough: The transient intense coughing fit that occurs in some users immediately after trenbolone injection (more commonly with Acetate than Enanthate) is not fully mechanistically characterised. The most credible hypothesis involves oil embolisation, tiny oil droplets from the injection entering the bloodstream and reaching the pulmonary vasculature, triggering prostaglandin release and local irritation. This causes intense bronchospasm lasting 30–90 seconds, typically resolving completely. It is frightening and occasionally severe enough to cause brief loss of consciousness, but essentially harmless if brief. Users who experience frequent or prolonged tren cough should consider slower injection technique, warming the solution to reduce viscosity, and switching injection sites.
Cardiovascular strain: Trenbolone is among the most cardiovascularly demanding compounds in widespread use. Its combination of zero aromatization (eliminating estrogen’s cardioprotective effects on lipids and endothelial function), high androgenicity (directly contributing to LDL rise, HDL suppression, and erythropoiesis), and possible direct myocardial AR activation contributes to a uniquely unfavourable cardiovascular risk profile. Blood pressure elevation with trenbolone is common and can be substantial (10–20 mmHg systolic in some users), requiring active management with antihypertensives. The cardiovascular case against extended or high-dose trenbolone use is strong.
Neurological and psychological effects: Trenbolone reliably produces mood alteration in a proportion of users, increased aggression, irritability, anxiety, and in some users, depression, paranoia, and acute psychological crisis. The mechanisms underlying these effects involve multiple systems: glucocorticoid receptor agonism in brain regions including the amygdala and hippocampus, the dopaminergic effects of progesterone receptor activation (which suppresses dopaminergic tone), and possibly direct effects on neurosteroid pathways. These are not merely psychological responses to being on something, they are pharmacologically mediated and vary in severity with individual receptor sensitivity and dose. Users who notice personality changes, relationship strain, or mood symptoms that are clearly correlated with trenbolone use should treat this as a compound contraindication, not a management problem.
Dose Creep and Compound Accumulation
Dose creep is one of the most pervasive patterns in advanced performance pharmacology, and it is pharmacologically predictable rather than simply a matter of poor judgment. It proceeds in recognisable steps.
First, a user establishes a baseline protocol that produces good results and acceptable side effects. Over time, the anabolic response to that protocol diminishes, not because the protocol has stopped working, but because the user’s natural set point of body composition adaptations has shifted toward the new normal, and the perceived rate of progress slows. The subjective experience is that the protocol is not doing anything anymore, which is typically incorrect, it is maintaining the adaptations achieved, which requires equivalent hormonal stimulus, not increasing stimulus.
Second, the user adds a compound or increases a dose to try to recapture the original rate of progress. This produces a temporary boost, which is then followed by adaptation to the new level. Side effects also increase at the new level, typically subtly at first, such that the user attributes them to other factors.
Third, a new side effect emerges that is actually caused by a recently added compound. Rather than removing the compound, the user adds an ancillary to manage the side effect. This ancillary creates its own interactions and side effects. The protocol has now expanded in both directions, more anabolic compounds and more management compounds, while the actual anabolic benefit has not increased proportionally.
The deeper pharmacology is messier than the bro claim that “the receptor is fully saturated, so nothing else matters.” There is no clean universal AR ceiling that can be read off a forum chart and applied to every tissue and every protocol. Receptor occupancy, co-regulator availability, local enzyme environment, free-androgen fraction, and non-AR pathways all matter. But the practical conclusion still points in one direction: marginal returns fall much faster than users want to believe once androgen exposure is already very high. Past that point, dose escalation usually buys a worse side-effect ratio, not a proportionally better anabolic result.
What the Research Shows About Cardiovascular Risk
The cardiovascular research on long-term AAS use has matured substantially over the past decade, and the evidence is concerning in ways that advanced users should understand precisely rather than reactively.
Left ventricular hypertrophy (LVH) is the best-documented structural cardiac change from AAS use. Long-term androgen exposure produces concentric LVH, thickening of the left ventricular wall disproportionate to cavity volume, in a dose- and duration-dependent manner. This is distinct from the eccentric hypertrophy of exercise-induced cardiac adaptation (increased cavity volume with proportional wall thickening). Concentric LVH impairs diastolic function and is associated with increased arrhythmia risk. Cross-sectional studies comparing long-term AAS users to age-matched controls show larger average LV wall thickness and impaired diastolic function in the user group even after controlling for exercise volume.
Coronary artery disease: Accelerated coronary atherosclerosis in AAS users is documented by imaging studies showing greater coronary artery calcium scores and plaque burden in long-term users compared to age-matched non-users. The mechanism involves the combination of sustained HDL suppression, LDL elevation, endothelial dysfunction from androgen-mediated nitric oxide suppression, and systemic inflammation from polypharmacy. The data does not suggest that all AAS users will develop coronary artery disease, it suggests that the risk trajectory is accelerated, and that the cumulative effect of multiple cycles over years is not zero.
The harm-reduction implication is clear: cycle duration, total lifetime compound exposure, and inter-cycle recovery time are the primary modifiable risk factors. A user who completes 8 weeks of a carefully managed cycle per year, ensures lipid and hematocrit normalisation between cycles, and monitors cardiovascular markers annually is at fundamentally lower risk than a user running blast and cruise protocols year-round regardless of how carefully individual cycles are managed. Time off compounds allows HDL recovery, hematocrit normalisation, and partial reversal of some structural cardiac changes, these benefits are measurable and clinically significant.
When to Take a Break: Recovery Cycles and Long-Term Thinking
The decision framework for recovery cycles is under-developed in most advanced-user discourse because it requires engaging honestly with the long-term risk data rather than focusing exclusively on short-term performance optimisation. The evidence basis for meaningful time off is clear and supported by multiple lines of data.
A meaningful recovery period, defined as sufficient time off all exogenous androgens for lipid normalisation, hematocrit normalisation, HPTA recovery to at least a functional level, and partial reversal of LVH, requires a minimum of 12–16 weeks for most users following moderate cycles, and potentially 6–12 months following extended high-dose protocols. The rule of thumb “time on equals time off” is a reasonable minimum for moderate users; it is likely insufficient following extended blast and cruise protocols.
The markers that confirm adequate recovery have been normalised: LH and FSH have returned to at least 50% of baseline; total testosterone is at or above pre-cycle baseline; HDL has recovered to within 15% of pre-cycle baseline; hematocrit is below 48%; blood pressure is at or below pre-cycle levels.
Users who find themselves unable or unwilling to take meaningful recovery periods, because the physical and psychological effects of being off-cycle are intolerable, should recognise this as a dependence signal rather than a training necessity. AAS dependence is a documented clinical phenomenon with endocrine, psychological, and behavioural dimensions, and the recognition of it does not require meeting formal addiction criteria. It requires only honesty about whether pharmacology is serving the training goal or the training goal has become the pretext for ongoing pharmacology.
Selected references for major clinical, mechanistic, or protocol claims. Community-practice points may not be cited individually.