GH Secretagogues, hGH, and Practical Outcomes
A mechanistic breakdown of GH secretagogue pathways, CJC-1295 DAC vs. non-DAC pharmacokinetics, ipamorelin vs. GHRP-2, GHRP-6 and Hexarelin, MK-677 long-term effects, exogenous hGH dosing logic, how lifters actually talk about BPC-157 and TB-500, and the risks of insulin co-administration.
- Explain the GHRH and GHRP pathways and how they differ mechanistically
- Select between CJC-1295 DAC and non-DAC based on pharmacokinetic goals
- Compare Ipamorelin, GHRP-2, GHRP-6, and Hexarelin on cortisol, appetite, and GH pulse characteristics
- Evaluate what MK-677 actually does over long-term use and its metabolic tradeoffs
- Understand exogenous hGH dosing logic and how to confirm IGF-1 response
- Explain why BPC-157 and TB-500 built such a strong reputation and where they tend to fit in real-world use
- Describe the mechanism and dangers of insulin co-administration
The GH Secretagogue System: GHRH vs. GHRP Pathways
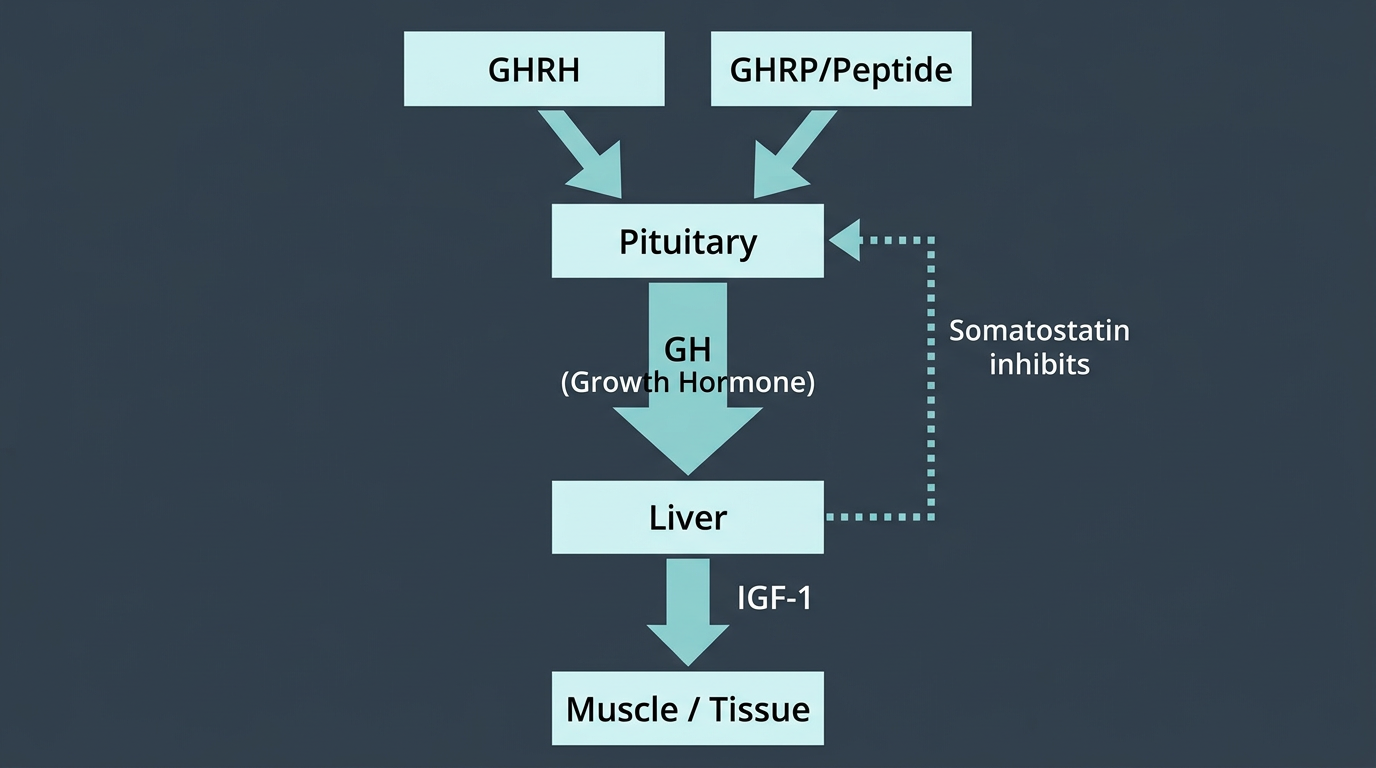
Growth hormone secretion from the anterior pituitary is regulated by two primary stimulatory pathways and one inhibitory pathway. Understanding which pathway each peptide engages is essential for rational protocol design, and for understanding what these compounds actually do versus what is commonly claimed about them.
The first stimulatory pathway is the GHRH pathway. GHRH is a 44-amino-acid peptide secreted from the hypothalamus that binds the GHRH receptor on somatotroph cells in the anterior pituitary, activating adenylyl cyclase, raising intracellular cAMP, and triggering GH synthesis and release. GHRH stimulation produces a physiological GH pulse, but its effect is gated by somatostatin, the inhibitory signal that defines the pulsatile rhythm of GH secretion. When somatostatin tone is high, GHRH stimulation produces little to no GH release. This is the critical constraint on GHRH-mimetic peptides: CJC-1295 and other GHRH analogues only produce meaningful GH secretion when administered during a somatostatin trough.
The second stimulatory pathway is the ghrelin/GHS-R1a pathway. Ghrelin is an endogenous peptide primarily produced in the stomach that binds the GHS-R1a receptor in the pituitary and hypothalamus. This receptor pathway stimulates GH release via a mechanism partially independent of somatostatin gating, ghrelin-receptor agonists can blunt somatostatin’s inhibitory effect and trigger GH release even outside the physiological trough. As a result, GHRP-class compounds (GHRP-2, Ipamorelin) produce GH release more reliably across varying administration times than GHRH-class compounds.
The third pathway is somatostatin (SRIF), released from the hypothalamus to inhibit both GH secretion and IGF-1 production in the liver. Somatostatin is the gatekeeper: no GH-stimulating signal produces meaningful GH output when somatostatin tone is high. That is also why the combination of a GHRH analogue + a GHRP-class compound is synergistic rather than merely additive: the GHRP-class compound suppresses somatostatin gating, allowing the GHRH-class compound to act on primed somatotrophs. The combined GH pulse is substantially larger than either alone.
The practical design implication: for maximum GH pulse amplitude, co-administer CJC-1295 (non-DAC) + Ipamorelin approximately 30–45 minutes before bed (or post-workout), when natural somatostatin tone is lower. This protocol produces meaningful GH pulses that translate to elevated IGF-1 over time.
CJC-1295: DAC vs. Non-DAC: Half-Life and Pulse Architecture
CJC-1295 exists in two distinct forms with fundamentally different pharmacokinetic profiles, and the naming convention is unfortunately confusing in many product listings.
Non-DAC CJC-1295, often sold as Modified GRF 1-29 (Mod-GRF 1-29), is a GHRH analogue with a half-life of approximately 30 minutes in vivo. It produces a sharp, physiologically-timed GH pulse when injected, mimicking the natural GHRH pulse pattern. Because it clears rapidly, it does produce tonic GH stimulation. Each injection triggers one pulse, then the compound is eliminated. This pulse-based stimulation preserves pituitary responsiveness and produces an IGF-1 profile more similar to physiological GH patterns.
CJC-1295 with DAC (Drug Affinity Complex), the true original CJC-1295, has a half-life of approximately 6–8 days due to a lysine-DAC modification that allows covalent albumin binding, dramatically extending its circulation time. DAC-modified CJC-1295 produces sustained GHRH receptor stimulation rather than pulsatile stimulation. The result is continuous low-amplitude GH stimulation rather than discrete pulses. Some practitioners argue this leads to greater IGF-1 elevation over time; others argue it desensitises GHRH receptors and disrupts the physiological pulsatile pattern that the GH axis depends on for receptor recycling and maintained pituitary sensitivity.
The practical consensus in the performance pharmacology community is that non-DAC (Mod-GRF 1-29) + Ipamorelin is preferred for its physiological pulse-mimicking pattern, cleaner IGF-1 response, and preserved pituitary sensitivity. DAC-modified CJC-1295 has the convenience of less frequent dosing but at the cost of physiological pulse architecture. If the goal is convenience and consistent IGF-1 elevation without concern for pulse architecture, DAC may be acceptable; if the goal is maximising GH pulse amplitude and preserving long-term pituitary responsiveness, non-DAC is the rational choice.
Ipamorelin vs. GHRP-2: Cortisol, Appetite, and GH Pulse Quality
Both Ipamorelin and GHRP-2 are synthetic ghrelin-receptor agonists, but they differ significantly in selectivity and off-target effects.
GHRP-2 is a potent GHS-R1a agonist that produces substantial GH pulses, among the largest of the GHRP class, but selectivity for GH release is limited. GHRP-2 also significantly elevates cortisol and prolactin, likely via direct pituitary and adrenal stimulation through the ghrelin receptor pathway. At typical performance doses (100–200 mcg per injection), cortisol elevation with GHRP-2 can be 30–40% above baseline. For users during cutting phases or recovery periods where cortisol management is a priority, this is a meaningful disadvantage. GHRP-2 also stimulates appetite via ghrelin-receptor pathways in the hypothalamus, for users in a caloric deficit, this appetite stimulation can be a significant adherence problem.
Ipamorelin is a far more selective GHS-R1a agonist. It produces GH pulses of similar or slightly smaller amplitude to GHRP-2 but with minimal cortisol or prolactin elevation at therapeutic doses, and with substantially less appetite stimulation. This selectivity makes Ipamorelin the preferred GHRP for most performance applications, particularly when used during cutting phases, in users managing stress or cortisol-driven recovery issues, or in women (who are more sensitive to cortisol and prolactin perturbation). The selectivity of Ipamorelin is its primary clinical advantage and the main reason it has largely replaced GHRP-2 in informed performance peptide protocols.
The combination protocol, Ipamorelin 100–200 mcg + Non-DAC CJC-1295 100 mcg, administered 2–3 times daily or at minimum before sleep, produces GH pulses that, over several months of consistent use, reliably elevate IGF-1 levels, improve sleep quality (via GH-mediated SWS enhancement), improve body composition, and support collagen synthesis and connective tissue recovery. The timeline for measurable IGF-1 elevation is typically 4–8 weeks of consistent twice-or-more-daily administration, single-injection-per-day protocols produce meaningfully less IGF-1 elevation.
GHRP-6 and Hexarelin: Where They Fit
GHRP-6 is one of the older, messier GHRPs. It does raise GH, but what most people remember about it is the hunger. For some users that is the whole reason to use it in a gaining phase. For anyone trying to stay lean or keep appetite under control, it gets old fast. Compared with Ipamorelin, it is a rougher peptide with more appetite and less finesse. Compared with GHRP-2, most people think of it as a little less punchy for GH release but more likely to make you want to eat everything in sight.
Hexarelin sits at the harder-edged end of the class. It is potent, and that is why it still gets brought up, but it also has more baggage. Long runs raise more concern about tolerance, and it does not have the same “easy to live with” feel that made Ipamorelin popular. If someone wants the strongest GHRP pulse possible, Hexarelin usually enters the conversation. If someone wants something they can actually run without a lot of friction, it usually drops back out.
In practice, most modern protocols drift toward Ipamorelin or skip the older GHRPs completely. GHRP-2 still has a place if someone wants a stronger hit and accepts the extra mess. GHRP-6 is mostly for people who specifically want the hunger effect. Hexarelin is the one people read about, respect, and then often decide they do not really need.
MK-677: The Oral Secretagogue: Long-Term Effects and Metabolic Tradeoffs
MK-677 (ibutamoren) is an orally active, non-peptide GHS-R1a agonist with a half-life of approximately 24 hours, allowing once-daily oral administration. It produces sustained GH and IGF-1 elevation, not through pulses but through tonic ghrelin-receptor stimulation. This distinguishes it from peptide-based secretagogues and makes it pharmacologically unique in the GH-axis modulation space.
The clinical evidence base for MK-677 is actually more substantial than for most peptides in this category, with controlled trials in healthy young men, growth-hormone-deficient adults, and elderly populations demonstrating consistent IGF-1 elevation at 25 mg/day. GH pulse amplitude is increased, and total 24-hour GH secretion rises significantly. The IGF-1 response is reliable and measurable within 2–4 weeks.
The metabolic tradeoffs of MK-677 are important and frequently underemphasised. As a ghrelin-receptor agonist, MK-677 shares ghrelin’s appetite-stimulating properties, dramatically so at 25 mg/day. Many users report voracious appetite, which is useful in bulking contexts and actively problematic in cutting contexts. Insulin sensitivity is reduced with MK-677, the compound raises fasting glucose and impairs insulin sensitivity in a dose-dependent manner. In users with baseline metabolic dysfunction or who are using the compound at high doses (above 25 mg), this can progress to measurable insulin resistance and impaired glucose tolerance. Long-term, continuous use at 25 mg/day consistently worsens metabolic markers in a way that shorter peptide protocols do not. Water retention from IGF-1 and GH elevation is also common, particularly in the first 4–8 weeks of use.
One smaller point that is easy to miss: chronic ghrelin-receptor stimulation is not just a growth-and-appetite story. There are preclinical signals that long-term MK-677-type stimulation may influence fear-related learning, salience, or emotional tone. That is nowhere near as well-established as the glucose issue, and it is not enough to say MK-677 “causes anxiety” in human users. But if someone says a long MK-677 run made them feel oddly edgy, more stress-reactive, or just mentally off, that report is not crazy on its face.
Prolactin sits in a similar category. It is not the headline issue with MK-677, and it is not something I would expect most users to notice before appetite, edema, or fasting-glucose drift. But ghrelin-receptor signaling is happening in a pituitary environment where hormone systems overlap. So if someone on long-term MK-677 picks up mild prolactin-related symptoms or an unexplained lab bump, MK-677 is at least worth keeping in the picture instead of assuming it is hormonally neutral.
The practical recommendation is to treat MK-677 as a tool for specific contexts, extended bulking periods where appetite stimulation is an asset and the user accepts metabolic monitoring obligations, rather than a maintenance compound run year-round. Fasting glucose and insulin sensitivity should be monitored quarterly with long-term use.
Exogenous hGH: Dosing Logic, IGF-1 Confirmation, and the Carpal Tunnel Problem
Exogenous hGH (recombinant human growth hormone, somatropin) directly replaces or augments pituitary GH output. Unlike secretagogues that stimulate endogenous GH release, exogenous hGH delivers GH regardless of somatostatin tone, pituitary responsiveness, or natural pulsatile rhythm. This makes its IGF-1 response more predictable and dose-proportional than secretagogue-based approaches.
Dosing in performance contexts ranges widely. Bodybuilders traditionally use 2–4 IU/day for body composition and recovery effects, with doses up to 8–10 IU/day for dramatic body composition changes in contest preparation contexts. The dose-response for IGF-1 elevation is not linear, there is meaningful individual variation in hepatic IGF-1 production in response to GH, and some users require higher doses to achieve similar IGF-1 targets.
The primary method to confirm that exogenous hGH is actually working is IGF-1 testing. Many users are surprised to find their IGF-1 has not elevated substantially on doses they expected to be effective, this can result from underdosed product (a serious and common quality control problem), poor injection timing, or individual variation in GH-to-IGF-1 conversion efficiency. Draw an IGF-1 level after 4–6 weeks of consistent dosing to confirm response. Target IGF-1 levels for performance use are generally in the upper third of the normal range (approximately 200–350 ng/mL for most adult assays).
The side effect profile of hGH at performance doses is dominated by edema (water retention in soft tissues) and carpal tunnel syndrome, the latter from direct IGF-1-mediated connective tissue swelling that compresses the median nerve in the carpal tunnel. Carpal tunnel symptoms from hGH are dose-dependent and typically reversible with dose reduction or cessation. Other common sides include joint swelling, morning stiffness, and in long-term high-dose use, acromegalic features. Visceral fat organ hypertrophy is a legitimate concern with years of high-dose use, and insulin co-administration plays a larger role than GH alone in this phenomenon.
BPC-157 and TB-500
BPC-157 (Body Protection Compound 157) is a synthetic pentadecapeptide derived from a protein found in gastric juice. TB-500 is a synthetic peptide fragment of thymosin beta-4, an endogenous protein involved in actin polymerisation and cellular migration.
BPC-157 (Body Protection Compound 157) is one of the few peptides that has a genuinely strong reputation in anecdotal use. People use it for angry elbows, beat-up shoulders, cranky knees, tendon insertions, gut issues, and the kind of nagging soft-tissue problems that are hard to train around. TB-500 is usually treated as the broader add-on when the issue feels more systemic, older, or spread across multiple spots.
That reputation did not come out of nowhere. The animal work on BPC-157 is broad and points in the same direction over and over: better tendon healing, better ligament healing, better gut healing, and faster recovery in several injury models. Proposed mechanisms include more VEGF-driven angiogenesis, better fibroblast activity, and lower inflammatory signaling around damaged tissue. TB-500 makes more sense as a migration-and-repair peptide, with animal work centered on wound healing, angiogenesis, inflammation control, and tissue remodeling through its [actin] effects.
What matters in practice is that the anecdotal pattern is unusually consistent. Across forums, logs, coaches, and long-time peptide users, the same story keeps showing up: BPC-157 is the one people actually feel. The tissue calms down, pain drops, range of motion improves, and training becomes tolerable again. TB-500 is less often the star on its own, but it gets used constantly as the “broader recovery” add-on when someone wants more than a local peptide.
The real practical caution here is not “nobody thinks these work.” Plenty of people do. The real problems are product quality, fake or underdosed vials, and unrealistic expectations. These peptides do not replace load management, rehab, or time. They tend to look best when there is a clear soft-tissue problem, inflammation is a real part of the picture, and the user is also fixing the thing that caused the issue.
GH and Insulin Co-Administration: Nutrient Partitioning and Hypoglycemia Danger
The combination of exogenous hGH with insulin is the most pharmacologically potent body composition protocol in existence and the most dangerous routine PED combination. Understanding the mechanism of both the benefit and the danger is essential for any advanced user in this space.
The nutrient partitioning rationale is as follows. hGH is lipolytic and anti-insulin, it reduces peripheral glucose uptake and upregulates fatty acid mobilisation. Administered peri-workout with insulin, the GH’s IGF-1-mediated protein synthesis stimulation and the insulin’s anabolic signaling (via PI3K/Akt/mTOR) combine to create a dramatically enhanced anabolic environment in skeletal muscle. Insulin drives glucose and amino acids into muscle cells; GH upregulates protein synthesis machinery and satellite cell activation via IGF-1; the combination produces greater lean mass accretion than either alone.
The hypoglycemia danger is severe and non-trivial. Insulin lowers blood glucose by driving cellular uptake. Exogenous insulin, whether fast-acting analogues like Humalog Insulin and Novolog Insulin, the intermediate Humulin R, or long-acting Lantus Insulin, has a dose-dependent blood glucose-lowering effect that does not plateau at any dose within the performance-use range. Hypoglycemia (blood glucose below 70 mg/dL) produces cognitive impairment, confusion, seizure, and at severe levels, death. The margin between a performance dose of fast-acting insulin and a fatal overdose is narrow, especially in the absence of adequate carbohydrate intake timed to insulin action.
The critical safety rules for any insulin use: never inject insulin without fast-acting carbohydrates immediately available (50+ grams consumed within 15 minutes of injection for fast-acting analogues); never inject insulin and then sleep (you will not wake up from severe nocturnal hypoglycemia); never inject long-acting insulin without a clear plan for 12–24 hour glucose management; always have a glucagon emergency kit available. Insulin is a life-saving medication that kills when misused. It has no place in performance enhancement.
Selected references for major clinical, mechanistic, or protocol claims. Community-practice points may not be cited individually.